Innovative technology driven solutions for Biotech and Pharma
MyBiotech is a CDMO offering end to end development, manufacturing services and innovative products for biotech and pharma.
At MyBiotech innovative technologies are part of the solution bringing your project one step closer to success.
MyBiotech has fermentation capacities for all kinds of microorganisms.
MyBiotech uses its proprietary method and equipment for cell rupture.
Our extraction processes are tailor-made for high efficiency and product yield.
MyBiotech produces natural Omega-3 fatty acids from algae. We can specifically produce both DHA- and DHA/EPA-rich oil.
MyPharma
We offer a broad range nano and micro particle formulations.
TECHNOLOGY TRANSFER
Technology transfer services for your established nanoparticle and microparticle formulations.
FORMULATION
Solutions for pharmaceuticals with a special focus on low soluble substances.
REGISTRATION
We can help you with your export and registration projects europe wide.
MyBiotech is a member of Instillo group which consolidates and supplies administrative support to an independent group of companies that operates over 20 countries world wide. It has established a strong network and built up a competitive team offering their expert knowledge for a number of national and international companies.

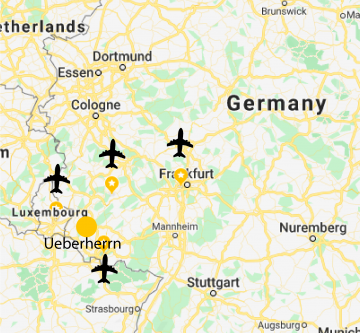
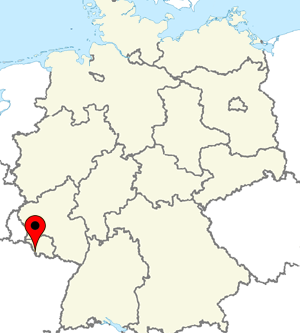
MyBiotech is located in Ueberherrn. The facility is a 45 minutes drive from Luxemburg airport and 90 minutes drive from Frankfurt am Main airport enabling easy access to global airports.
latest EVENTS & news
from the blog
MJR PharmJet and MyBiotech are combined in merger of equals. The company will combine the best of the pharmaceutical and biotechnology worlds. Combined company will drive technology oriented product innovation addressing the needs of the customers. The new group will trade under the MyBiotech banner.
